|
|
Case Report
| ||||||
| Metformin associated lactic acidosis complicates orthotopic liver transplantation | ||||||
| Andrew William Pool1, Zoka Milan2 | ||||||
|
1MBChB FRCA, ST7 Anaesthesia, King's College Hospital, Denmark Hill, London UK.
2MD, PhD, FRCA, FCIM, Consultant Anaesthetist, Honorary Senior Lecturer, Visiting Professor, King's College Hospital, London, UK. | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article |
| Pool AW, Milan Z. Metformin associated lactic acidosis complicates orthotopic liver transplantation. Edorium J Anesth 2016;2:28–32. |
|
Abstract
|
|
Introduction:
We present a case of severe lactic acidosis during orthotopic liver transplant (OLT) in a 71-year-old male taking metformin for type 2 diabetes mellitus (DM). The early and unexpectedly severe lactic acidosis can be explained by metformin associated lactic acidosis (MALA), a rare and serious side effect of metformin usage not previously described during liver transplantation.
Case Report: A 71-year-old male with NASH cirrhosis and hepatocellular carcinoma presented for OLT with a background of hypertension and type 2 diabetes mellitus and estimated Glomerular Filtration Rate of 74. He received a donation after cardiac death (DCD) organ from a 60-year-old with an 8-hour cold ischemic time. Despite an uneventful anesthetic induction and relative cardiovascular stability, an unexpectedly severe lactic acidosis with pH 7.2 and a lactate of 9.7 mmol/L developed in the late dissection phase. This continued to worsen requiring intra-operative continuous veno-venous hemofiltration and prolonged ventilatory and inotropic support. The liver graft functioned satisfactorily. Conclusion: Though lactic acidosis is common during liver transplant, the rapid onset and severity prior to the anhepatic phase despite relative cardiovascular stability is unusual. This suggests impaired lactate clearance, a key feature of MALA. The MALA is a rare condition with prevalence estimated at between one and five cases per 100,000. The mainstay of treatment is organ support and hemofiltration. Unexpected severe lactic acidosis in the absence of more common causes should lead clinicians to consider MALA, particularly in patients with underlying renal impairment. | |
|
Keywords:
Liver transplant, Lactic acidosis, Metformin
| |
|
Introduction
| ||||||
|
Nonalcoholic fatty liver disease (NASH) has become the most common cause of liver disease worldwide with an estimated prevalence of 20% in the US [1] [2] [3]. The spectrum disease of disease ranges from mild macroscopic changes to NASH cirrhosis and hepatocellular carcinoma [4]. Risk factors include central obesity, hyperglycemia, type 2 diabetes, arterial hypertension and hypertriglyceridemia. This means a significant number of patients presenting with NASH cirrhosis for OLT will be on treatment for type 2 diabetes mellitus. Metformin, a dimethylbiguanide, is a widely used oral antihyperglycemic drug used in the long-term treatment of type 2 diabetes mellitus. It is contraindicated in severe renal dysfunction and should be used with caution in severe renal disease [5]. Metformin associated lactic acidosis (MALA) is a rare complication of metformin with an estimated prevalence if 1-5/100000 and mortality up to 50% [6]. It has not previously been reported during liver transplantation. We present a case of an unexpectedly severe lactic acidosis during orthotopic liver transplantation (OLT) for NASH. | ||||||
|
Case Report
| ||||||
|
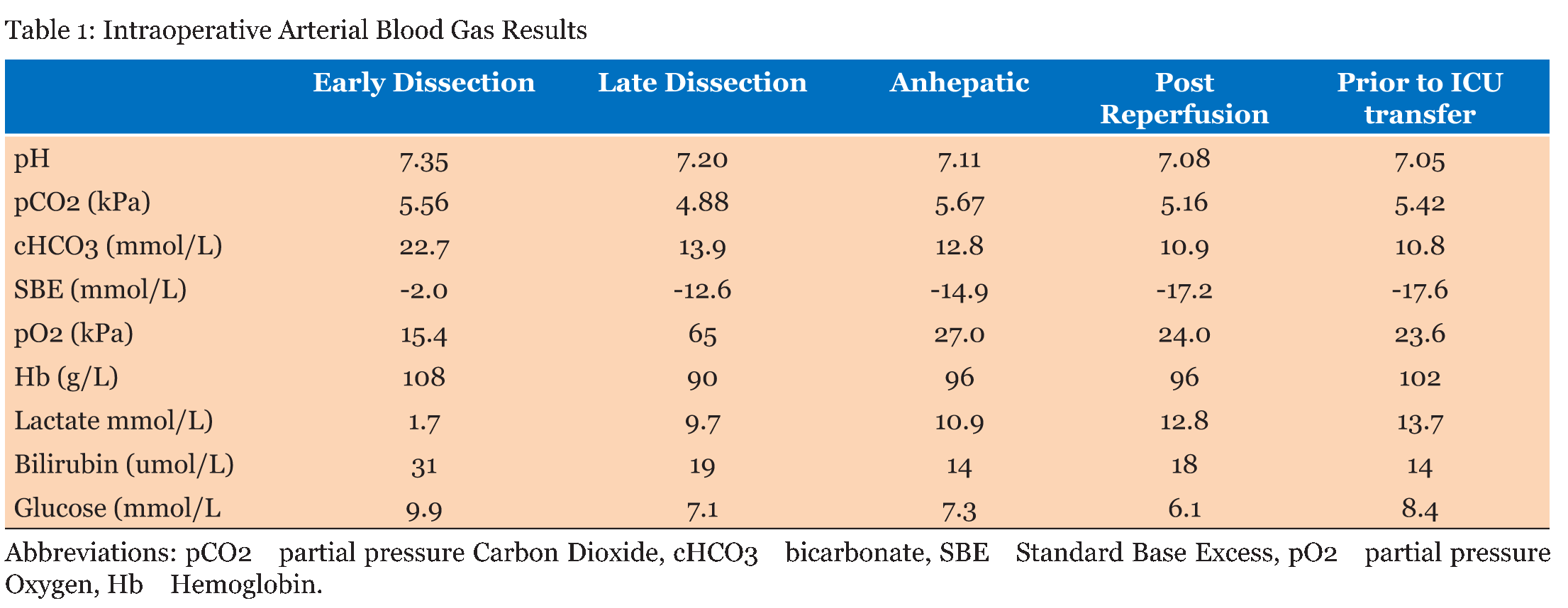
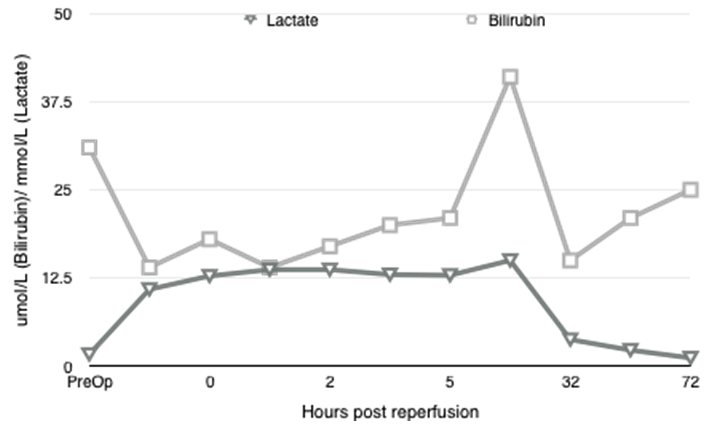
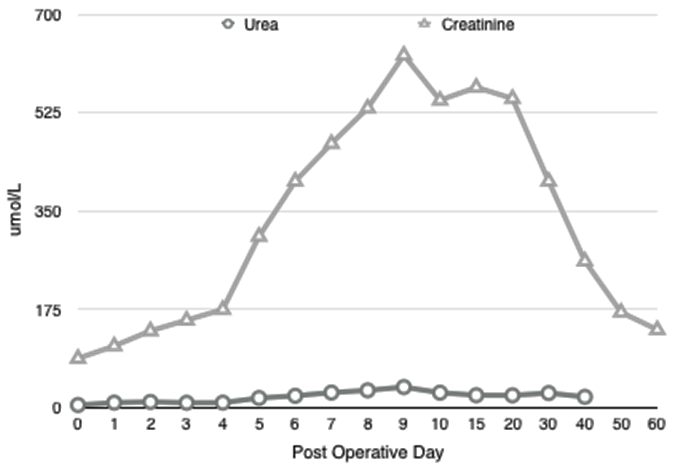
A 71-year-old blood group A+ve gentleman presented for OLT on a background of NASH cirrhosis and hepatocellular carcinoma for which he had undergone a TACE procedure one year previously. The NASH was diagnosed after an acute jaundiced episode three years previously with no associated portal hypertension or encephalopathy. Child Pugh B. UKELD score was 51. He had a background of hypertension and diabetes mellitus type 2, diagnosed eight years previously. His medications were novomix insulin, metformin 1 g BD, gliclazide and ramipril. He had also recently been treated with prednisolone for possible temporal arteritis, stopping 2 days prior to transplant. Cardiopulmonary exercise testing (CDX) showed moderate, severe impairment of VO2 max (71% of predicted) and an anaerobic threshold of 10.2ml/kg/min. The interpretation of the results suggested musculoskeletal de-conditioning. An operative risk score C (high risk) was recorded. Cardiac echo demonstrated no valvular lesions and good left ventricular function. Estimated glomerular filtration rate was 74 ml/min preoperatively. He was listed for a donor liver and three months later a transplant organ became available. The donor liver was a DCD organ from a 60-year-old with an eight hour cold ischemic time. The Cincinnati donor risk index for liver transplantation calculator gives a risk score of 2.6, which represents a high risk donor organ. It should however be noted that this tool is only fully validated for the North American population [7]. The patient had taken his last dose of metformin approximately 14 hours prior to surgery. Anesthetic induction preceded uneventfully and arterial monitoring, central venous and dialysis line were placed. Cardiac output monitoring (LidcoRapid, Lidco ltd) and depth of anesthesia monitoring (BIS, Covidien) was instigated. Isoflurane was used for maintenance of anesthesia alongside infusions of calcium (4 mmol/hr), atracurium (40 mg/hr) and fentanyl. The dissection phase was three hours long. Cardiovascular stability was maintained, with mean arterial pressures of 65 mmHg targeted. It required a noradrenaline infusion at 0.05 µg/kg/min. There were no prolonged periods of hypotension. The heart rate increased slowly from 80 post induction to 100 bpm, with a brief response to fluid challenges. Urine output during dissection was 205 ml (under 0.5 ml/kg/hr.) Thromboelastography revealed a slightly prolonged R time that was treated with fresh frozen plasma (FFP). Moderate blood loss was treated initially with two liters of colloid (Gelofusin), then three units of packed red cells and three units of FFP. The noradrenaline infusion was increased to 0.3 µg/kg/minute. The arterial blood gas result in the late dissection phase (Table 1) showed a dramatic increase in lactate levels with no obviously identifiable reason. The anion gap was raised at 21, consistent with lactic acidosis, despite apparently adequate oxygenation and oxygen delivery indicated on cardiac output monitoring. There was a brief period of hypotension on clamping of the IVC but this responded well to fluid boluses and vasopressors. The noradrenaline infusion was further increased to 0.5 mg.kg.min. The anhepatic phase lasted 60 minutes, with lactate levels increasing slightly (Figure 1). Sodium bicarbonate 100 mmol was given prior to reperfusion due to worsening acidosis. The liver was transplanted using a piggyback technique with a bile duct to duct anastomosis. Cardiovascular stability during re-perfusion was achieved with calcium, metaraminol boluses and sequential declamping from the surgical team and no prolonged hypotension occurred. In the face of worsening metabolic acidosis, continuous veno-venous hemofiltration (CVVH) was started in theatre during bile duct anastomosis and closure. Urine output was only 100 ml from re-perfusion to ICU transfer four hours later. Total blood loss was estimated at 4.2 L. The postoperative course was prolonged, with 10 days of hemofiltration and inotropic support and prolonged ventilatory support. The acute kidney Injury was prolonged and the patient required regular hemodialysis as an inpatient for a further 2 weeks before returning to an eGFR of 60 which remained stable on discharge (Figure 2). Liver USS on day-1 showed acceptable flows in the vessels in the postoperative period. There was no evidence of biliary duct dilatation and 5x4 cm hematoma adjacent to the porta hepatis. Plasma metformin levels were not taken, but in the context of massive transfusion and use of CVVH, plasma levels are unlikely to be representative. | ||||||
| ||||||
| ||||||
| ||||||
|
Discussion
| ||||||
|
We described a case of patient who underwent OLT with a sudden severe lactic acidosis which, in the absence of more common causes, can be explained by MALA. Lactic acidosis is a common issue during liver transplant with multiple contributing factors including the patient's pre-morbid state, significant intraoperative blood loss and reperfusion injury. What is unusual in this case is that the onset of severe lactic acidemia occurred prior to the anhepatic phase and after a straightforward dissection phase with only moderate blood loss and relative cardiovascular stability. This suggests impairment of lactate clearance, one of the key features of MALA. Metformin, a dimethylbiguanide, is a widely used oral antihyperglycemic drug used in the long-term treatment of type 2 diabetes mellitus. It has an absolute oral bioavailability of 50–60% and gastrointestinal absorption is apparently complete within six hours. Metformin is not metabolized and 90% of absorbed metformin is eliminated unchanged via the urine. Plasma protein binding is negligible, thus it can be removed by dialysis. The antihyperglcaemic effect of Metformin is thought to occur via increased glucose transport into cells and decreased hepatic gluconeogenesis. It decreases the activity of pyruvate dehydrogenase and the transport of mitochondrial reducing agents, and thus enhances anaerobic metabolism even in the presence of adequate oxygen. Inhibition of pyruvate dehydrogenase then channels the conversion of pyruvate into lactate rather than the aerobic pathway [8]. Studies have shown it to be effective in reducing both hyperglycemia and the microvascular complications of diabetes [9]. It is contraindicated in patients with an eGFR of <30 and prescribed with caution in eGFR 30–45 or those at risk of sudden deterioration in renal function as the risk of lactic acidosis is increased. It should also be withdrawn or avoided in patients with suspected tissue hypoxia. The risk/benefit should be discussed with patients with mild/moderate hepatic or cardiac impairment [5]. Lactic acidosis is subdivided into two types; type A from the accumulation of lactate from glycolysis during anaerobic respiration and type B where increased lactate production and reduced clearance causes an imbalance leading due lactic acidosis. The MALA is a form of type B lactic acidosis [10]. The MALA is a rare condition with prevalence estimated at between one and five cases per 100,000. Though rare, it has a reported mortality of 30–50% [11]. A number of confounding factors make accurate estimation difficult. Most case control studies exclude off label usage of metformin, thus removing the potentially higher risk patient group [12]. Additionally, the lack of measurement of plasma metformin levels means cases may be missed as the patient is treated for other causes of lactic acidosis. Formal diagnosis requires serum metformin levels, a test not routinely performed in most centers. Clinicians require a high index of suspicion and a marked lactic acidosis in the absence of hypoxia in a patient on metformin should lead to the consideration of MALA as a possible diagnosis. The mainstay of treatment is organ supportive therapy and hemofiltration as metformin is removed by dialysis [13] [14]. Perioperative guidance from NICE and the drug manufacturers suggests withdrawal of metformin prior to surgery though NICE themselves admit the evidence is limited [15][16]. Given the unplanned and time-critical nature of donor liver transplant, it is unlikely that it is feasible to withhold more than a single dose of metformin prior to surgery. | ||||||
|
Conclusion
| ||||||
|
Metformin associated lactic acidosis (MALA) is a rare condition and clinicians require a high index of suspicion MALA has not previously been reported during liver transplant and making a definitive diagnosis in the face of multiple potential causes of lactic acidosis is challenging. However, when unexpectedly severe lactic acidosis occurs in the absence of more common causes, we would urge clinicians to consider MALA, particularly in patients with underlying renal impairment. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Andrew William Pool – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Zoka Milan – Substantial contributions to conception and design, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2016 Andrew William Pool et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|